
Drug Companies on Trial: The Shocking Cases That Made Headlines
For decades, famous drug lawsuits involving pharmaceutical giants and their products have been making headlines. These cases often highlight important issues such as drug safety, efficacy, and marketing practices and have significant implications for both the pharmaceutical industry and public health.
In this article, we will explore some of the most high-profile drug lawsuits in recent history and discuss their impact on the industry and society as a whole.
The Opioid Crisis and Purdue Pharma

The opioid epidemic is one of the most severe and devastating drug-related crises that the United States has experienced in recent years. This crisis has caused the tragic deaths of thousands of people and resulted in countless cases of addiction across the country. Unfortunately, the opioid epidemic is often linked to the practices of Purdue Pharma, the manufacturer of OxyContin, and the pharmaceutical industry as a whole.
The situation became alarming in 2020, with the number of reported deaths due to opioid overdoses surging to 68,630, which was a significant increase from the previous years. This number continued to rise in 2021, with a total of 80,411 reported overdose deaths. These statistics paint a bleak picture of the opioid epidemic and underscore the need for immediate and effective solutions.
Purdue Pharma has faced numerous lawsuits related to its marketing practices, which allegedly downplayed the risk of addiction associated with its products. The company has paid billions of dollars in settlements and fines related to these lawsuits, and its owners have faced criminal charges. This case highlights the importance of drug safety and ethical marketing practices.
Tepezza Hearing Loss Lawsuit and Horizon Therapeutics
The Tepezza hearing loss lawsuit has put Horizon Therapeutics, the manufacturer of the prescription drug, in the spotlight. Tepezza is used to treat thyroid eye disease but has been linked to hearing loss in some patients, leading to several lawsuits against the company. A significant number of patients experienced side effects, which included hearing loss symptoms, with 10% of patients affected.
It’s important to note that the drug was approved by the FDA for treating thyroid eye disease, and the potential side effects should be weighed against the benefits of the treatment.
The Vioxx Scandal and Merck
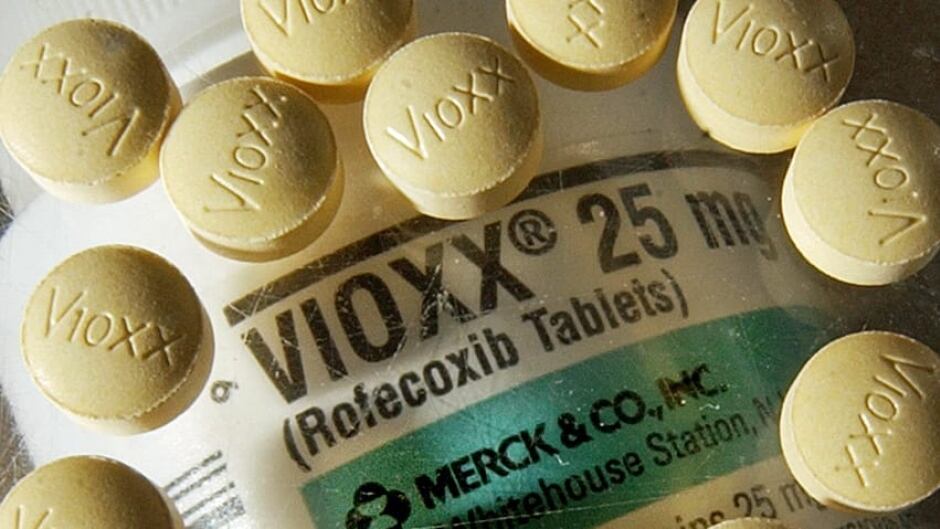
In 1999, Merck & Co., one of the largest pharmaceutical companies in the world, introduced a new painkiller called Vioxx. The drug was marketed as a safe and effective treatment for chronic pain, with the advantage of causing fewer gastrointestinal side effects than other pain medications. The drug quickly became a blockbuster, with sales reaching over $2 billion in 2003.
However, in 2004, a study found that Vioxx increased the risk of heart attacks and strokes in patients who used the drug for an extended period. This prompted Merck to voluntarily withdraw the drug from the market in September of that year, after an estimated 80 million people had taken it.
The withdrawal of Vioxx and the subsequent discovery of its potential risks led to a wave of lawsuits against Merck. Plaintiffs claimed that the company had failed to adequately warn patients and doctors about the risks associated with the drug, and that they had been harmed as a result of taking it.
The Vioxx lawsuits were among the largest and most high-profile drug lawsuits in history, with over 27,000 individual cases filed against Merck in the United States alone. The company ultimately paid out settlements totaling over $4 billion to resolve the lawsuits, making it one of the largest drug settlements in history.
Johnson & Johnson and Talcum Powder Lawsuits

Johnson & Johnson, a renowned company, has been embroiled in a series of lawsuits linked to their talcum powder products. These lawsuits allege that the use of the company’s talcum powder increases the risk of ovarian cancer in women. Unfortunately, this is a serious health concern, and Johnson & Johnson has paid billions of dollars in settlements related to these lawsuits.
Many research studies involving thousands of women have been conducted to investigate the link between talcum powder and ovarian cancer. According to studies, women who have used talcum powder are at a 30% higher risk of being diagnosed with ovarian cancer compared to women who have not used talcum powder. These findings are alarming and highlight the need for greater scrutiny of the safety of personal care products.
These cases highlights the importance of being vigilant about potential side effects and risks associated with prescription drugs. It also underscores the need for pharmaceutical companies to prioritize patient safety and conduct thorough testing and research before bringing products to market.
